
![]()

The Hydroxyapatite synthetic bone substitute, NEOBONE, a new functional synthetic bone filler, is a solution to the common problems of all existing bone filler materials. Based on the notion that a necessary requirement of a bone filler is its ability to allow biological tissue to grow speedily inside the grafted area (newly generated bones), it has a porous structure that facilitates the tissue to penetrate the structure and aids new tissue formation.
NEOBONE has a porous structure
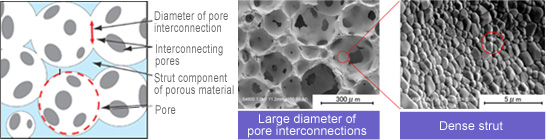
In principal, studies (animal tests) of NEOBONE's bone conduction ability, the rapid growth of the biological tissue into the specimen and more favorable bone conduction (manifestation of new bone formation capability and exhibition of strength) compared with the conventional synthetic bone substitutes were confirmed. Also, in clinical exercises, it is expected to demonstrate rapid bone formation and strength. NEOBONE also offers a number of benefits for patients, such as a significant reduction in physical and emotional stress during recuperation, reduced medical costs, and shorter rehabilitation periods.
 In order to allow penetration of biological tissues into the bone structure, if the porosity becomes higher and the interconnecting pore size larger, the strength of the synthetic bone decreases markedly in the conventional synthetic bone substitute.
In order to allow penetration of biological tissues into the bone structure, if the porosity becomes higher and the interconnecting pore size larger, the strength of the synthetic bone decreases markedly in the conventional synthetic bone substitute.
The frame portion of the pore of NEOBONE is made of very dense sintered hydroxyapatite, and so has enough strength in handling during grafting operations, or against mechanical loads. At the same time, it has excellent machinability, which enables it to process/machine at the time of operations.

Since NEOBONE has excellent biocompatibility and adequate strength that results in easy handling, it has a potentially higher scope for applications in fields such as regenerative medicine and as scaffolds in tissue engineering.
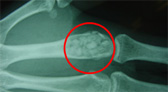
In the principal tests (animal tests), the biological tissues's rapid penetration into the NEOBONE r structure and the bone conduction ability are confirmed, and its porous structure is proved effective. Also, in the clinical tests, where the product was used for various cases, no inflammation was observed after the grafting operations. In more than half the cases, X-ray evaluations revealed that bone development occurred in the grafted areas within a short span of two months, which demonstrated the safe and excellent bone conduction properties of NEOBONE.
NEOBONE products are distributed by Aimedic MMT Co., Ltd.
Aimedic MMT Co., Ltd.
Kokumin-Kaikan Sumitomo-Seimei Bldg. 4F
1-2, Otemae 2-chome, Chuo-ku, Osaka City, Osaka 540-0008, Japan
TEL +81-6-6941-8255 FAX +81-6-6941-8251
e-mail: asamori@aimedicmmt.co.jp